Selenium
Selenium is a chemical element; it has symbol Se and atomic number 34. It has various physical appearances, including a brick-red powder, a vitreous black solid, and a grey metallic-looking form. It seldom occurs in this elemental state or as pure ore compounds in Earth's crust. Selenium (from σελήνη 'moon') was discovered in 1817 by Jöns Jacob Berzelius, who noted the similarity of the new element to the previously discovered tellurium (named for the Earth). Selenium is found in metal sulfide ores, where it substitutes for sulfur. Commercially, selenium is produced as a byproduct in the refining of these ores. Minerals that are pure selenide or selenate compounds are rare. The chief commercial uses for selenium today are glassmaking and pigments. Selenium is a semiconductor and is used in photocells. Applications in electronics, once important, have been mostly replaced with silicon semiconductor devices. Selenium is still used in a few types of DC power surge protectors and one type of fluorescent quantum dot. Although trace amounts of selenium are necessary for cellular function in many animals, including humans, both elemental selenium and (especially) selenium salts are toxic in even small doses, causing selenosis.[11] Symptoms include (in decreasing order of frequency): diarrhea, fatigue, hair loss, joint pain, nail brittleness or discoloration, nausea, headache, tingling, vomiting, and fever.[12] Selenium is listed as an ingredient in many multivitamins and other dietary supplements, as well as in infant formula, and is a component of the antioxidant enzymes glutathione peroxidase and thioredoxin reductase (which indirectly reduce certain oxidized molecules in animals and some plants) as well as in three deiodinase enzymes. Selenium requirements in plants differ by species, with some plants requiring relatively large amounts and others apparently not requiring any.[13] CharacteristicsPhysical properties Selenium forms several allotropes that interconvert with temperature changes, depending somewhat on the rate of temperature change. When prepared in chemical reactions, selenium is usually an amorphous, brick-red powder. When rapidly melted, it forms the black, vitreous form, usually sold commercially as beads.[14] The structure of black selenium is irregular and complex and consists of polymeric rings with up to 1000 atoms per ring. Black selenium is a brittle, lustrous solid that is slightly soluble in CS2. Upon heating, it softens at 50 °C and converts to gray selenium at 180 °C; the transformation temperature is reduced by presence of halogens and amines.[15] The red α, β, and γ forms are produced from solutions of black selenium by varying the evaporation rate of the solvent (usually CS2). They all have a relatively low, monoclinic crystal symmetry (space group 14) and contain nearly identical puckered cyclooctaselenium (Se8) rings as in sulfur.[16] The eight atoms of a ring are not equivalent (i.e. they are not mapped one onto another by any symmetry operation), and in fact in the γ-monoclinic form, half the rings are in one configuration (and its mirror image) and half in another.[17][18] The packing is most dense in the α form. In the Se8 rings, the Se–Se distance varies depending on where the pair of atoms is in the ring, but the average is 233.5 pm, and the Se–Se–Se angle is on average 105.7°. Other selenium allotropes may contain Se6 or Se7 rings.[15] The most stable and dense form of selenium is gray and has a chiral hexagonal crystal lattice (space group 152 or 154 depending on the chirality)[19] consisting of helical polymeric chains, where the Se–Se distance is 237.3 pm and Se–Se–Se angle is 103.1°. The minimum distance between chains is 343.6 pm. Gray selenium is formed by mild heating of other allotropes, by slow cooling of molten selenium, or by condensing selenium vapor just below the melting point. Whereas other selenium forms are insulators, gray selenium is a semiconductor showing appreciable photoconductivity. Unlike the other allotropes, it is insoluble in CS2.[15] It resists oxidation by air and is not attacked by nonoxidizing acids. With strong reducing agents, it forms polyselenides. Selenium does not exhibit the changes in viscosity that sulfur undergoes when gradually heated.[14][20] IsotopesSelenium has seven naturally occurring isotopes. Five of these, 74Se, 76Se, 77Se, 78Se, 80Se, are stable, with 80Se being the most abundant (49.6% natural abundance). Also naturally occurring is the long-lived primordial radionuclide 82Se, with a half-life of 8.76×1019 years.[10] The non-primordial radioisotope 79Se also occurs in minute quantities in uranium ores as a product of nuclear fission. Selenium also has numerous unstable synthetic isotopes ranging from 64Se to 95Se; the most stable are 75Se with a half-life of 119.78 days and 72Se with a half-life of 8.4 days.[21] Isotopes lighter than the stable isotopes primarily undergo beta plus decay to isotopes of arsenic, and isotopes heavier than the stable isotopes undergo beta minus decay to isotopes of bromine, with some minor neutron emission branches in the heaviest known isotopes.
Chemical compoundsSelenium compounds commonly exist in the oxidation states −2, +2, +4, and +6. It is a nonmetal (more rarely considered a metalloid) with properties that are intermediate between the elements above and below in the periodic table, sulfur and tellurium, and also has similarities to arsenic.[15] Chalcogen compounds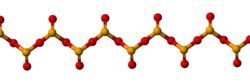 Selenium forms two oxides: selenium dioxide (SeO2) and selenium trioxide (SeO3). Selenium dioxide is formed by combustion of elemental selenium:[14] Se + O2 → SeO2
It is a polymeric solid that forms monomeric SeO2 molecules in the gas phase. It dissolves in water to form selenous acid, H2SeO3. Selenous acid can also be made directly by oxidizing elemental selenium with nitric acid:[24] 3 Se + 4 HNO3 + H2O → 3 H2SeO3 + 4 NO
Unlike sulfur, which forms a stable trioxide, selenium trioxide is thermodynamically unstable and decomposes to the dioxide above 185 °C:[14][24] 2 SeO3 → 2 SeO2 + O2 (ΔH = −54 kJ/mol)
Selenium trioxide is produced in the laboratory by the reaction of anhydrous potassium selenate (K2SeO4) and sulfur trioxide (SO3).[25] Salts of selenous acid are called selenites. These include silver selenite (Ag2SeO3) and sodium selenite (Na2SeO3). Hydrogen sulfide reacts with aqueous selenous acid to produce selenium disulfide: H2SeO3 + 2 H2S → SeS2 + 3 H2O
Selenium disulfide consists of 8-membered rings. It has an approximate composition of SeS2, with individual rings varying in composition, such as Se4S4 and Se2S6. Selenium disulfide has been used in shampoo as an antidandruff agent, an inhibitor in polymer chemistry, a glass dye, and a reducing agent in fireworks.[24] Selenium trioxide may be synthesized by dehydrating selenic acid, H2SeO4, which is itself produced by the oxidation of selenium dioxide with hydrogen peroxide:[26] SeO2 + H2O2 → H2SeO4
Halogen compoundsSelenium reacts with fluorine to form selenium hexafluoride: Se8 + 24 F2 → 8 SeF6
In comparison with its sulfur counterpart (sulfur hexafluoride), selenium hexafluoride (SeF6) is more reactive and is a toxic pulmonary irritant.[27] Selenium tetrafluoride is a laboratory-scale fluorinating agent. The only stable chlorides are selenium tetrachloride (SeCl4) and selenium monochloride (Se2Cl2), which might be better known as selenium(I) chloride and is structurally analogous to disulfur dichloride. Metastable solutions of selenium dichloride can be prepared from sulfuryl chloride and selenium (reaction of the elements generates the tetrachloride instead), and constitute an important reagent in the preparation of selenium compounds (e.g. Se7). The corresponding bromides are all known, and recapitulate the same stability and structure as the chlorides.[28] The iodides of selenium are not well known, and for a long time were believed not to exist.[29] There is limited spectroscopic evidence that the lower iodides may form in bi-elemental solutions with nonpolar solvents, such as carbon disulfide[30] and carbon tetrachloride;[29] but even these appear to decompose under illumination.[31] Some selenium oxyhalides—seleninyl fluoride (SeOF2) and selenium oxychloride (SeOCl2)—have been used as specialty solvents.[14] Metal selenides Analogous to the behavior of other chalcogens, selenium forms hydrogen selenide, H2Se. It is a strongly odiferous, toxic, and colorless gas. It is more acidic than H2S. In solution it ionizes to HSe−. The selenide dianion Se2− forms a variety of compounds, including the minerals from which selenium is obtained commercially. Illustrative selenides include mercury selenide (HgSe), lead selenide (PbSe), zinc selenide (ZnSe), and copper indium gallium diselenide (Cu(Ga,In)Se2). These materials are semiconductors. With highly electropositive metals, such as aluminium, these selenides are prone to hydrolysis, which may be described by this idealized equation:[14]
Alkali metal selenides react with selenium to form polyselenides, Se2− Other compoundsTetraselenium tetranitride, Se4N4, is an explosive orange compound analogous to tetrasulfur tetranitride (S4N4).[14][33][34] It can be synthesized by the reaction of selenium tetrachloride (SeCl4) with [((CH Selenium reacts with cyanides to yield selenocyanates:[14]
Organoselenium compoundsSelenium, especially in the II oxidation state, forms a variety of organic derivatives. They are structurally analogous to the corresponding organosulfur compounds. Especially common are selenides (R2Se, analogues of thioethers), diselenides (R2Se2, analogues of disulfides), and selenols (RSeH, analogues of thiols). Representatives of selenides, diselenides, and selenols include respectively selenomethionine, diphenyldiselenide, and benzeneselenol. The sulfoxide in sulfur chemistry is represented in selenium chemistry by the selenoxides (formula RSe(O)R), which are intermediates in organic synthesis, as illustrated by the selenoxide elimination reaction. Consistent with trends indicated by the double bond rule, selenoketones, R(C=Se)R, and selenaldehydes, R(C=Se)H, are rarely observed.[36] History Selenium (Greek σελήνη selene meaning "Moon") was discovered in 1817 by Jöns Jacob Berzelius and Johan Gottlieb Gahn.[37] Both chemists owned a chemistry plant near Gripsholm, Sweden, producing sulfuric acid by the lead chamber process. Pyrite samples from the Falun Mine produced a red solid precipitate in the lead chambers, which was presumed to be an arsenic compound, so the use of pyrite to make acid was discontinued. Berzelius and Gahn, who wanted to use the pyrite, observed that the red precipitate gave off an odor like horseradish when burned. This smell was not typical of arsenic, but a similar odor was known from tellurium compounds. Hence, Berzelius's first letter to Alexander Marcet stated that this was a tellurium compound. However, the lack of tellurium compounds in the Falun Mine minerals eventually led Berzelius to reanalyze the red precipitate, and in 1818 he wrote a second letter to Marcet describing a newly found element similar to sulfur and tellurium. Because of its similarity to tellurium, named for the Earth, Berzelius named the new element after the Moon.[38][39] In 1873, Willoughby Smith found that the electrical conductivity of grey selenium was affected by light.[40][41] This led to its use as a cell for sensing light. The first commercial products using selenium were developed by Werner Siemens in the mid-1870s. The selenium cell was used in the photophone developed by Alexander Graham Bell in 1879. Selenium transmits an electric current proportional to the amount of light falling on its surface. This phenomenon was used in the design of light meters and similar devices. Selenium's semiconductor properties found numerous other applications in electronics.[42][43][44] The development of selenium rectifiers began during the early 1930s, and these replaced copper oxide rectifiers because they were more efficient.[45][46][47] These lasted in commercial applications until the 1970s, following which they were replaced with less expensive and even more efficient silicon rectifiers. Selenium came to medical notice later because of its toxicity to industrial workers. Selenium was also recognized as an important veterinary toxin, which is seen in animals that have eaten high-selenium plants. In 1954, the first hints of specific biological functions of selenium were discovered in microorganisms by biochemist, Jane Pinsent.[48][49] It was discovered to be essential for mammalian life in 1957.[50][51] In the 1970s, it was shown to be present in two independent sets of enzymes. This was followed by the discovery of selenocysteine in proteins. During the 1980s, selenocysteine was shown to be encoded by the codon UGA. The recoding mechanism was worked out first in bacteria and then in mammals (see SECIS element).[52] Occurrence Native (i.e., elemental) selenium is a rare mineral, which does not usually form good crystals, but, when it does, they are steep rhombohedra or tiny acicular (hair-like) crystals.[53] Isolation of selenium is often complicated by the presence of other compounds and elements. Selenium occurs naturally in a number of inorganic forms, including selenide, selenate, and selenite, but these minerals are rare. The common mineral selenite is not a selenium mineral, and contains no selenite ion, but is rather a type of gypsum (calcium sulfate hydrate) named like selenium for the moon well before the discovery of selenium. Selenium is most commonly found as an impurity, replacing a small part of the sulfur in sulfide ores of many metals. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||


