Cinnoline
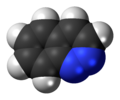
Cinnoline is an aromatic heterocyclic compound with the formula C8H6N2. It is isomeric with other naphthyridines including quinoxaline, phthalazine and quinazoline. Neither the unsubstituted heterocycle nor any derivatives is known to occur naturally.[3] PropertiesThe free base can be obtained as an oil by treatment of the hydrochloride with base. It co-crystallizes with one molecule of ether as white silky needles, (m.p. 24–25 °C or 75.2–77.0 °F) upon cooling ethereal solutions. The free base melts at 39 °C or 102.2 °F. It has a taste resembling that of chloral hydrate and leaves a sharp irritation for some time. Discovery and synthesisThe compound was first obtained in impure form by cyclization of the alkyne o-C6H4(N2Cl)C≡CCO2H in water to give 4-hydroxycinnoline-3-carboxylic acid. This material could be decarboxylated and the hydroxyl group reductively removed to give the parent heterocycle. This reaction is called the Richter cinnoline synthesis.[4] Improved methods exist for its synthesis. It can be prepared by dehydrogenation of dihydrocinnoline with freshly precipitated mercuric oxide. It can be isolated as the hydrochloride.[5] Cinnolines are cinnoline derivatives. A classic organic reaction for synthesizing cinnolines is the Widman–Stoermer synthesis,[6] a ring-closing reaction of an α-vinyl- aniline with hydrochloric acid and sodium nitrite:  A conceptually related reaction is the Bamberger triazine synthesis towards triazines. Another cinnoline method is the Borsche cinnoline synthesis. SafetyCinnoline is toxic.[3] See alsoReferences
|
||||||||||||||||||||||||||||||||||||||||||||||||||||